Introduction to Notch

Introduction to Notch
There are some antibodies:
1. Brontictuzumab
Recombinant Humanized (from mouse) antibody to Human Notch1.
2. Tarextumab
Recombinant human antibody to Human Notch2. Tarextumab (anti-Notch2/3, OMP-59R5) is a novel anti-cancer stem cell antibody identified by screening against the Notch3 receptor that binds to and prevents signaling through both the Notch2 and Notch3 receptors. Tarextumab has been shown in preclinical models to have broad-spectrum anti-tumor activity via inhibition of cancer stem cell growth, and promoting cell differentiation, as well as disrupting tumor angiogenesis by inhibiting vascular pericytes.
What is Notch?
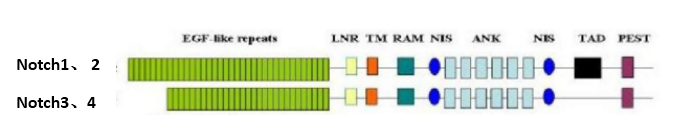
Figure1. Notch molecule
Notch gene was first found in Drosophila melanogaster in 1917. It was named for Notch because of its partial loss of function. Notch signaling pathway is a highly conserved cellular signaling system in most multicellular organisms. It is mainly composed of Notch, Notch ligand (DSL protein) and CSL (a class of DNA binding proteins). Mammals have four different Notch receptors, called Notch1, Notch2, Notch3 and Notch4. Notch receptor is a single channel transmembrane receptor protein. It is a heterogeneous oligomer composed of large extracellular groups, which binds to a smaller segment of Notch protein consisting of a short extracellular domain, a single transmembrane channel and a small intracellular domain in a calcium-dependent, non-covalent interaction.
From Drosophila remnant wings to targeted therapy for human tumors, Notch signaling pathway has been widely present and highly conserved in invertebrates and vertebrates, mediating the interaction of adjacent cells, regulating the differentiation and development of cells, tissues and organs. Notch pathway in mammals has complex and diverse functions, and is involved in important physiological processes such as stem cell regeneration and differentiation, hematopoiesis and T cell development. Mutations and abnormal expression of key molecules in Notch signaling are closely related to the occurrence and progression of various tumors, and different Notch receptors play different roles in the occurrence and development of tumors.
Structure
Notch signaling pathway is composed of Notch, Notch ligand (DSL protein) and CSL (a class of DNA binding proteins). Notch and its ligands are single transmembrane proteins.
Notch: It’s a protein with a molecular weight of about 300 KD. Drosophila has only one Notch gene and four in humans. Notch’s extracellular domain is a ligand-binding region with different numbers of EGF-like repeats (EGF-R) and three LNRs. The intracellular domain consists of RAM domain, 6 anchor protein repeats, 2 nuclear localization signals (NLS) and EST domain. RAM domain is the region that combines CSL, and PEST domain is related to Notch degradation.
Notch ligand: In Drosophila melanogaster, Notch ligand is Delta and Serrate, the nematode is Lag-2, and Notch ligand is also called DSL protein (Jagged in mammals). It is a single transmembrane glycoprotein. Its extracellular domain contains a number of EGF-like repeat regions, and the N-terminal has a DSL motif necessary for Notch binding.
CSL: A transcription factor called CBF1 in mammals. CSL recognizes and binds to a specific DNA sequence (GTGGGAA), which is located on the promoter of Notch-inducible gene. The target genes of Notch signal are alkaline helix-ring-helix transcription factors, which regulate the transcription of other genes directly related to cell differentiation.
Activation of Notch signaling pathway
The classical Notch signaling pathway is also called CBF1/RBP JK dependent pathway. CBF1/RBP-JK is a transcription inhibitor, which can specifically bind to DNA sequence CGTGGGAA and recruit SMRT, SKIP, I/II histone deacetylase proteins to form co-inhibitory complexes and inhibit the transcription of downstream genes. The activation of this pathway requires three steps of enzymatic digestion. After ribosome synthesis, the Notch receptor precursor protein encoded by Notch gene was cleaved by furin-like invertase at S1 site to produce two subunits, ECN and TM. ECN and TM are linked together by disulfide bonds to form a heterodimer Notch receptor located on the surface of the cell membrane. After ligand binding to the extracellular domain, Notch protein was cleaved into two fragments at S2 site under the action of metalloproteinase. N-terminal cleavage products (ECN) were phagocytized by ligand-expressing cells, while C-terminal cleavage products were further activated by Secretase protease complex at S3 site to release Notch protein-NICD. NICD into nucleus, it plays a role of signal transduction.
Regulation of Notch signaling pathway
Many intracellular and extracellular proteins can regulate Notch signals through various mechanisms, including intracellular proteins such as Delex and Numb, and extracellular proteins such as Fringe and Wingless. The regulation of Notch signaling pathway in the nucleus is a little complicated, which is related to many transcription factors and regulatory proteins, such as NF-KB, EMb-5, etc. At the same time, Notch signaling pathway is also regulated by ubiquitination-related proteins and self-feedback. Notch signaling pathway also plays an important role in regulating apoptosis and proliferation of multicellular organisms. Studies have shown that TNF signaling can activate Notch 2 and up-regulate its expression, and then induce apoptosis by regulating the key regulators of apoptosis.
Functions of Notch signaling pathway
Notch signaling pathway is an important way of intercellular communication. It participates in the gene regulation mechanism that controls the process of multicellular differentiation in embryos and adults. Notch signals also play a role in the following processes:
Neuronal function and development; stability and angiogenesis of differentiation fate of arterial endothelial cells; regulation of key intercellular communication between endocardium and myocardium in the process of valve formation, ventricular development and differentiation; maintenance of heart valve homeostasis, affecting human diseases involving cardiovascular system; timely differentiation of cell lines to differentiate endocrine and exocrine pancreas; influence cell duality Decisions of fate, such as affecting the choice of differentiation fate between intestinal secretion and absorption lines, assisting the expansion of hematopoietic stem cell chambers during bone development and participating in the formation of osteoblast lineages, imply the potential therapeutic role of Notch in bone regeneration and osteoporosis, the proliferation of vascular endothelial cells, the commitment of T-cell lineages from common lymphoid precursors, and several others. The same developmental stage determines the fate of breast cells; some non-nuclear mechanisms may be involved, such as the control of actin cytoskeleton by tyrosine kinase Abl.
Notch transduction is a complex signaling pathway in mammals. It not only participates in the differentiation, proliferation and apoptosis of cancer stem cells, but also plays an important role in the invasion of cancer. As an important regulator of cell proliferation and metabolism, this pathway is closely dependent on the interaction between tumor microenvironment and other signaling pathways. How to effectively identify and target Notch-related participants and target molecules for different tumors is very important for anti-cancer therapy. Targeted therapy for cancer in the future has great research prospects in drug inhibitors, monoclonal antibodies and gene level.